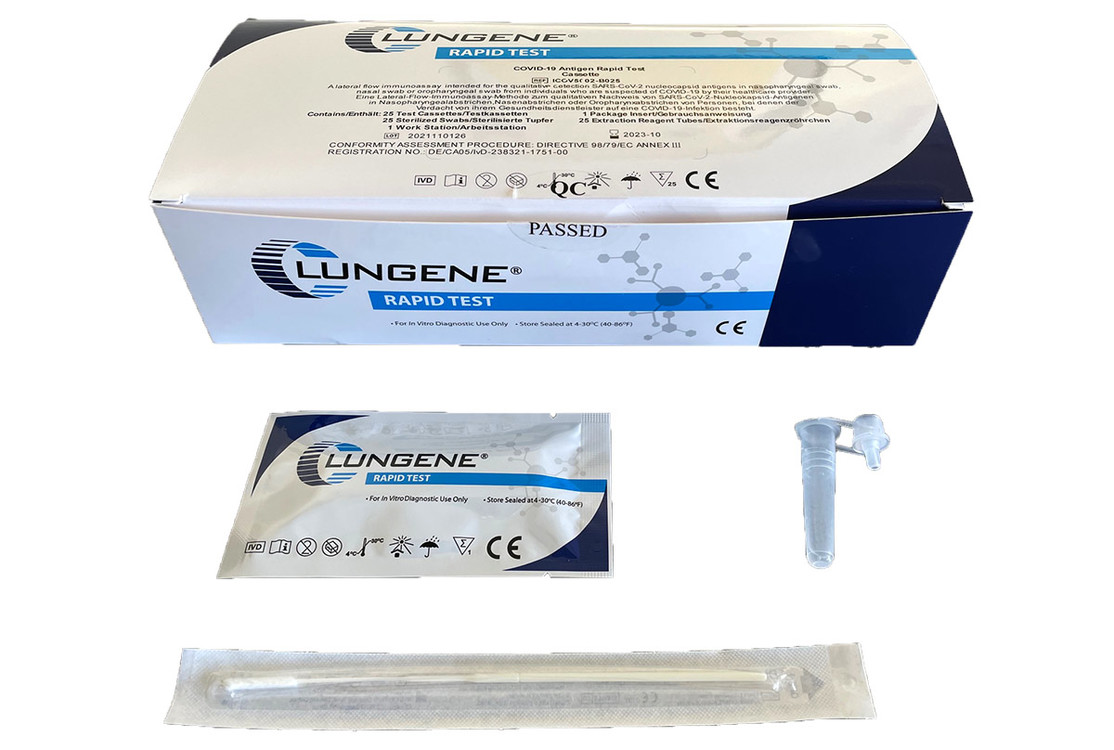
The new rapid test from Hangzhou Clungene Biotech Co. Ltd detects SARS-CoV-2 virus antigens in a nasopharyngeal, pharyngeal or nasal sample.
EOF code: 2830000664890
• Contains positive and negative control samples
• Clinical susceptibility> 98% for SARS-CoV-2
compared to a reference molecular test
• Clinical specialty> 99.7% for SARS-CoV-2
compared to a reference molecular test
• Result within 15 minutes
Each package contains:
25 Diagnostic Cassettes, 25 Extraction Reagents, 25 Sampling Tubes, 25 Saliva Collectors, 25 Sterile Swabs, 25 Droppers, 1 Packing Insert with Instructions for Use in Greek and 1 Workstation.
Orders
The product is not available to retail individuals. For more information contact Mr. Konstantinos Katsamoris at tel: 2651085858 or via e-mail at kkatsamoris@newpharm.gr.
The European Union Health
The European Union Health and Safety Committee on 18 February 2021 has approved a list of 16 rapid detection tests for Covid-19, the results of which are mutually recognized by all Member States and whose results are vital to facilitate cross-border traffic. cross-border contact tracking and treatment.
The 16 Approved Rapid Tests by the EU Health Insurance Committee
Technical Data Rapid Test Hangzhou Clungene Biotech Co (European Commision Database)
The effectiveness of the 16 tests on the list has been validated by an independent study by at least 1 EU Member State reference laboratory and their availability in at least 3 Member States effectively replaces the requirements of each Member State and is the roadmap difference from the hundreds of rapid tests. Covid-19 Antigen Detection with European Certification.
This list is the result of a unanimous decision by all the Member States of the European Union of 21 January 2021, based on the European Commission's proposal to establish a framework for the use of rapid antigen detection tests and the common recognition of COVID-19 results throughout its territory. The European Union.
EU Health Insurance Committee List
It should be noted that Greece must accept travelers who have been examined and have received a Certificate of Result based on the specific European list of approved fast Covid-19 antigen tests, and respectively those traveling from Greece to the European Union will present a similar Certificate of result.
Among the 16 Covid 19 rapid antigen detection tests included in this list is the Hangzhou Clungene Biotech Co. test. Ltd , one of the largest and most reliable biotechnology companies in China, which Newpharm A.E. represents for Greece and Cyprus.
We analyze below the technical specifications of the Covid 19 rapid antigen detection test of the company Hangzhou Clungene Biotech Co. Ltd
USEFUL LINKS
MANUFACTURER DOCUMENTS that include the following:
1) Clinical evaluation, validated by the Competent Government Authority of China, performed on 770 samples by selecting samples with symptoms within the first 7 days and asymptomatic suspected of being carriers of Covid 19, where the ability to detect the antigen test is confirmed 33 CT cycles and 37 CT cycles.
2) Reliability of the RAPID TEST CLUNGENE ANTIGONE, has been certified by independent laboratory evaluations in Germany, Belgium and Slovenia.
3) Confirmation of reliability RAPID TEST ANTIGONE CLUNGENE (Comparative evaluation of the sensitivity of rapid SARS-CoV-2 antigen tests).
4) List of the German Drug BfArM, indicating the sensitivity and specificity of the RAPID TEST CLUNGENE ANTIGEN
TECHNICAL SPECIFICATIONS CLUNGENE RAPID ANTIGON OF COVID 19
• Detects SARS-CoV-2 antigen
• Performed on a nasopharyngeal, pharyngeal or nasal sample
• It has CE-IVD approval
• It has all the necessary certificates from the European Authorities, EOF code: 2830000664890 and notification / approval by the Ministry of Health of Cyprus
• Produces results within 15 minutes
• Contains positive and negative control samples
• It has clinical sensitivity> 98% for SARS-CoV-2 compared to molecular reference test.
• It has clinical specificity> 99.7% for SARS-CoV-2 compared to molecular reference test
Detection limit 5.7 × 102TCID50 / mL
• There is an indication of the validity of the test in addition to the indication of positive or negative
• Long-term storage at room temperature (5 to 30 degrees Celsius)
• The execution and completion of the test is done exclusively with equipment available in the kit and the results are visually read without accompanying equipment
• Multiple samples can be performed simultaneously and independently - it is easy to use in the logic of Point of Care Tests so that no specialized experts in molecular biology experiments / tests are required, but it can be performed by health personnel (such as doctors, nursing staff).
• It is performed with disposable devices that are disposable and fully portable and with individually packed test devices and individual packaging of extraction buffer for each test.
the sensitivity / specificity information is documented by a kit study depending on the Ct of rtRT-PCR with which the comparison was made
- USEFUL LINKS
- TECHNICAL SPECIFICATIONS CLUNGENE RAPID ANTIGON OF COVID 19